Diabetes: Restoring glucose regulation through the eye

Research is a long-distance race full of challenges, according to Senior Professor Per-Olof Berggren. But if his innovative approach of transplanting insulin-releasing cells into the eye succeeds, he might just reach the finish line when it comes to diabetes treatments.
Per-Olof Berggren, today Senior Professor at the Rolf Luft Research Centre for Diabetes and Endocrinology, Department of Molecular Medicine and Surgery at Karolinska Institutet, has long studied diabetes and potential treatments. He began his research to gain a deeper understanding of what occurs in insulin-secreting cells under normal conditions, how they release insulin and regulate blood glucose levels, and why these processes fail in diabetes.
At first, he would never have thought of the idea of the eye as a transplantation site in people with diabetes, but soon after, he realized it could be possible. “The eye provides a supportive environment, and transplanted insulin-releasing cells survive well,” he says.
The cornea is a natural window into the body
Going back twenty years, Berggren and his team isolated insulin-secreting cells from mice, humans, rabbits, and rats and studied them outside the body. However, he recognized early on that this approach did not reflect what happens in a living organism.
“In isolation, the cells have no blood supply, no neural input, and no interaction with surrounding tissues. This led us to ask how we could study these cells inside a living organism while still being able to visualize what was happening inside them,” says Berggren. “That led us to use the anterior chamber of the eye as a transplantation site, with the cornea serving as a natural optical window for imaging.”
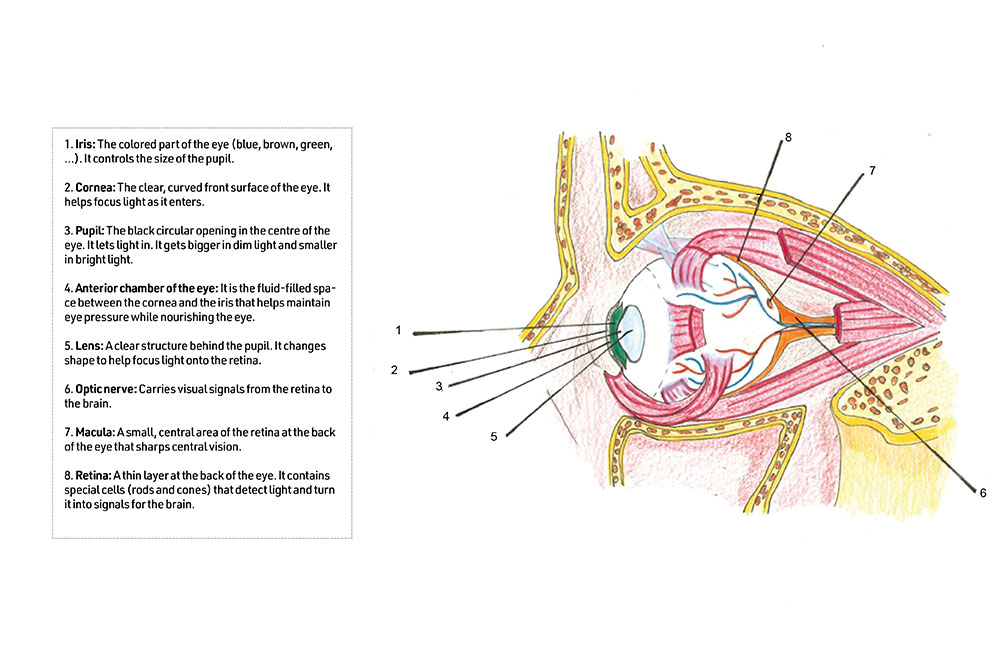
Transplanting these cells into the eye has several advantages, adds Berggren. “First, the cells can be visualized over time with advanced microscopy to assess their behavior, health, and function. Second, they are directly accessible, allowing local pharmacological treatment. Third, the cells can be removed if necessary. Finally, the eye is a relatively immune-privileged site, with less aggressive immune responses, particularly in the early phase after transplantation when the tissue is less vascularized.”
Transplanting insulin-secreting cells into the eye was a major conceptual shift in our research. It opened up entirely new possibilities, as we could now study many signals in great detail within a living organism.
When Berggren and his team transplanted insulin-secreting cells into the anterior chamber of the eye, the cells engrafted securely onto the iris, became well vascularized and innervated, and remained in place without migrating. These cells secrete insulin directly into the bloodstream in the same way they do in the pancreas. When blood glucose levels rise, glucose reaches the insulin-secreting cells in the eye, which respond to that increase by releasing insulin into the blood circulation. The resulting effect of insulin is therefore entirely normal and identical to the effect of insulin produced by the pancreas.
“Transplanting insulin-secreting cells into the eye was a major conceptual shift in our research. It opened up entirely new possibilities, as we could now study many signals in great detail within a living organism,” says Berggren. “Transplanting only 10% of the normal number of insulin-secreting pancreatic cells into the eye of a mouse is sufficient to cure its diabetes. When the same proportional calculation is applied to humans, there is enough space in the eye to theoretically achieve the same effect. In addition, the eye provides a highly supportive environment, as its fluid is rich in nutrients and oxygen, allowing transplanted cells to survive very well.”
The researchers have conducted these transplantation studies in monkeys and mice, and based on everything they observed, the animals retain normal vision. For this reason, they believe the procedure should also be safe for people with normal vision.
“The FDA indicated that if safety can be demonstrated in legally blind patients, there would be no reason not to extend the approach to patients with full vision,” Berggren says.

The clinical trials progress
Ten years ago, Berggren’s teamstarted a recruitment process to transplant insulin secreting cells in patients. Several admission criteria had to be met, which narrowed the number of candidates – they had to be legally blind and have a kidney transplant to avoid adding systemic immunosuppression.
Their first patient was transplanted in 2016, and despite the patient later dying from unrelated cardiovascular disease, the procedure itself was successful and showed new endogenous insulin production. However, the strict criteria made patient recruitment difficult, so the scientists revised the protocol with clinical specialists. Ophthalmologists pointed out that patients with corneal transplantation received systemic immunosuppression for a short period, which was then discontinued and replaced with local immunosuppression in the eye.
We are now moving forward, with one screened patient in Sweden awaiting transplantation in the US, another enrolled in China at West China Hospital in Chengdu, and several more patients in preparation.
This approach was much simpler for patients and far less burdensome and approval was obtained from the ethical committees including the FDA to adopt the new approach. But the process was halted by the COVID-19 pandemic after which the team had to rebuild the clinical infrastructure.

Per-Olof Berggren explains that research is a long-distance race full of challenges when experiments fail and have to be started over again.
But according to him, “the main challenge for any researcher is having the time and resources to pursue a question deeply, without expecting results immediately.”
“We are now moving forward, with one screened patient in Sweden awaiting transplantation in the US, another enrolled in China at West China Hospital in Chengdu, and several more patients in preparation. The study has also been expanded to include both insulin-dependent type 1 and type 2 diabetes patients,” Berggren adds.
And he strongly believes that initial transplantations will become routine within the next few years.
Engineering the future of diabetes treatment
Berggren’s research aims to refine the transplantation process and the cells themselves, not only to regulate blood glucose levels but ultimately to cure diabetes.
“Adopting a regenerative medicine approach based on replacing the dysfunctional cells with new ones is not enough; we want to make them more robust to immune and autoimmune attacks. Cells must be genetically modified. Strengthening these cells is essential for long-term therapeutic success,” he explains.
In the future, genetically engineered stem-cell-derived islets may offer a way to treat, and potentially cure, both type 1 and type 2 diabetes.
And he goes even further with a stem-cell strategy. “At the same time, we are developing stem-cell-derived pancreatic islets, and we have already established an effective protocol to generate them experimentally. In the future, genetically engineered stem-cell-derived islets may offer a way to treat, and potentially cure, both type 1 and type 2 diabetes.”
Given the global scale of diabetes, which affects hundreds of millions of people worldwide, future care must empower patients to become, to some extent, their own caregivers, supported by intelligent, self-monitoring therapeutic systems. So, Per-Olof and his team want to integrate advanced technologies, including electronics and optics.
“By developing micro-devices that can be implanted in the eye, we aim to deliver drugs to the transplanted tissue while simultaneously monitoring its function in real time. This approach could allow patients to take a more active role in managing their condition,” he says.
Taking responsibility
Diabetes is a very serious disease. Nowadays there’s still not currently a way to prevent or cure it, only treatments that manage symptoms. “Diabetes can lead to severe complications, including eye and cardiovascular problems, which makes it a critical area for research and healthcare investment,” Berggren says.
Which is why prevention must start early according to him. “Physical activity and health education should be prioritized in schools, so children learn from a young age to move, play, and stay active rather than spending all their time in front of screens,” he continues.
“It is important that people take responsibility for their own health. This does not mean extreme exercise or strict diets, but regular physical activity and a balanced, moderate diet. You can eat normal food and still live a full, healthy life if excess is avoided,” he concludes.
Updated: April 2, 2026, 06:53 am
Published: April 1, 2026












